- Blog
- Black diamond heavyweight screentap fleece gloves
- Random animal generator
- Firealpaca tips
- Restaurant busboy salary
- Shear brilliance
- The dungeon of naheulbeuk the amulet of chaos review
- Free downloadable audio clips for imovie
- Barcody tote carry
- Functional groups table
- Download mod hill climb racing apk
- Def elucidate
- Diffraction angle
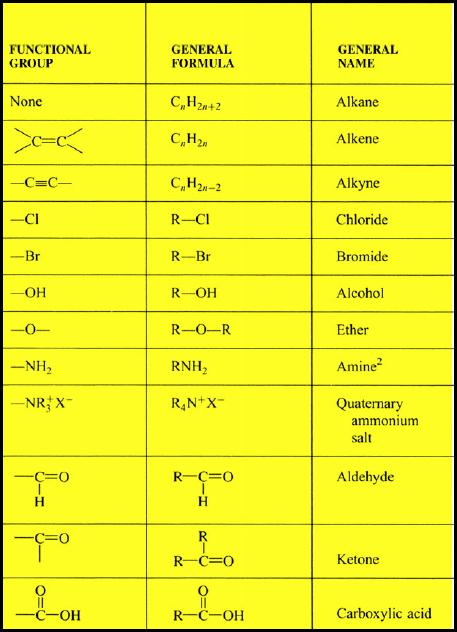
There can be more than one halogen substituted for a hydrogen atom on one haloalkane. Alcohols with shorter carbon chains are usually more soluble in water than those with longer carbon chains.Īlcohols tend to have higher boiling points than the hydrocarbons because of the strong hydrogen bond between hydrogen atoms of one hydroxyl group and the oxygen atom of another hydroxyl group. However, the carbon chain resists solubility, so there are two opposing trends in the alcohols. The hydroxyl group generally makes the alcohol molecule polar and therefore more likely to be soluble in water. The amount of the substance than can dissolve is the measure of its solubility. Solubility is a measure of the ability of a substance (solid, liquid or gas) to dissolve in another substance. Addition, Elimination and Substitution Reactions.Physical Properties and Branched Groups.Physical Properties and Functional Groups.Physical Properties and Intermolecular Forces.A carboxyl group (COOH) is a functional group consisting of a carbonyl group (C=O) with a hydroxyl group (O-H) attached to the same carbon atom. It contains the functional group C=O joined via carbon to another oxygen atom. Which functional group contains the functional group C=O joined via carbon? Functional groups can be used to distinguish similar compounds from each other. How do functional groups affect the reactivity of compounds?įunctional groups will undergo the same type of reactions regardless of the compound of which they are a part however, the presence of certain functional groups within close proximity can limit reactivity.

Therefore, it is only necessary to know about the chemistry of a few generic functions in order to predict the chemical behaviour of thousands of real organic chemicals.
The functional group approach ” works” because the properties and reaction chemistry of a particular functional group (FG) can be remarkably independent of environment. How does the functional group approach work? The same functional group will behave in a similar fashion, by undergoing similar reactions, regardless of the compound of which it is a part. In organic chemistry, a functional group is a specific group of atoms or bonds within a compound that is responsible for the characteristic chemical reactions of that compound. What are functional groups in organic chemistry? This compound is extensively used in the manufacture of phenol, aniline, and DDT as a solvent for paints and as a heat transfer medium. How is chloro benzene made?Ĭhlorobenzene is produced by chlorination of benzene in the presence of a catalyst, and is produced as an end product in the reductive chlorination of di- and trichlorobenzenes. They determine the characteristics and chemical reactivity of molecules. Functional groups are attached to the carbon backbone of organic molecules. They are responsible for the characteristic chemical reactions of those molecules. What is the purpose of functional groups?įunctional groups are specific groups of atoms or bonds within molecules. What are functional groups and why are they important? An organic compound is any compound that contains carbon and another element. They, therefore, determine the properties and chemistry of many organic compounds. Why are functional groups important?įunctional groups are important in chemistry because they are the portion of a molecule that is capable of characteristic reactions. The chloro group is a weak DMG and requires very low-temperature metallation conditions in order to prevent benzyne formation, for example, ortho-lithiated chlorobenzenes formed at −105☌ may be used in Michael reactions. Chlorobenzene is also used as a high-boiling solvent in the manufacture of adhesives, paints, paint removers, polishes, dyes, and drugs. The major use of chlorobenzene is as an intermediate in the production of commodities such as herbicides, dyestuffs, and rubber.